A Breakthrough in Lentiviral Vector Purification: Streamlining Suspension Feedstock Processing
Published date: 12 December 2023
Lentiviral vectors (LVVs) have garnered significant attention in the field of cell and gene therapy, offering a promising approach for treating a wide range of genetic diseases. However, the development of LVV-based therapeutic assets often encounters bottlenecks during both upstream and downstream processes, which can negatively impact pipeline milestones. One common challenge is the lengthy and complex sample preparation required to convert raw feed into purified substance, especially when dealing with suspension feedstock systems. These systems are favored for large-scale production due to scalability and cost-efficiency, but they come with high levels of contaminants, such as host cell proteins (HCP).
In response to these challenges, a ground-breaking, built for purpose, purification strategy has been developed, utilizing a novel spin column, the Nereus LentiHERO®. This innovative device features a functionalized high-flow, high-capacity composite nanofiber adsorbent called AstreAdept®.
AstreAdept® has previously demonstrated its ability to yield high LVV output from adherent production systems. The aim of this study was to optimize suspension feedstock clarification and compare the performance of Nereus LentiHERO® purification for both adherent and suspension LVV feedstocks.
Method
1. Experimental Plan: Adherent and suspension feedstocks were processed according to a specific experimental plan in Figure 1. Purification was carried out following the Nereus LentiHERO® NL100100 Technical User Guide.
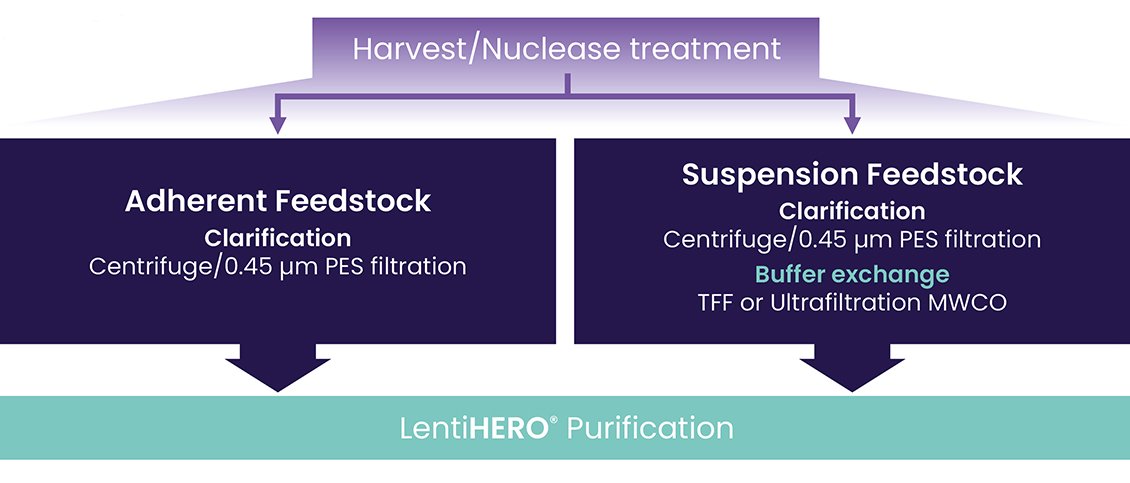
Figure 1.
2. Conditions for Clarification: Six conditions were tested to optimize the clarification step (table 1) by varying the centrifugation speed and size of MWCO filter. Condition #3 showed the highest TU titer, indicating minimal loss of functional LVV directly after this mid-stream process step. Here, the feed was clarified by centrifugation at 1500 x g, followed by a 100 kDa MWCO filtration. At this point in the purification process contaminants such as dsDNA and HCP will still need to be addressed, so further downstream purification is required to fully understand how the mid-stream process step fits into a representative workflow.
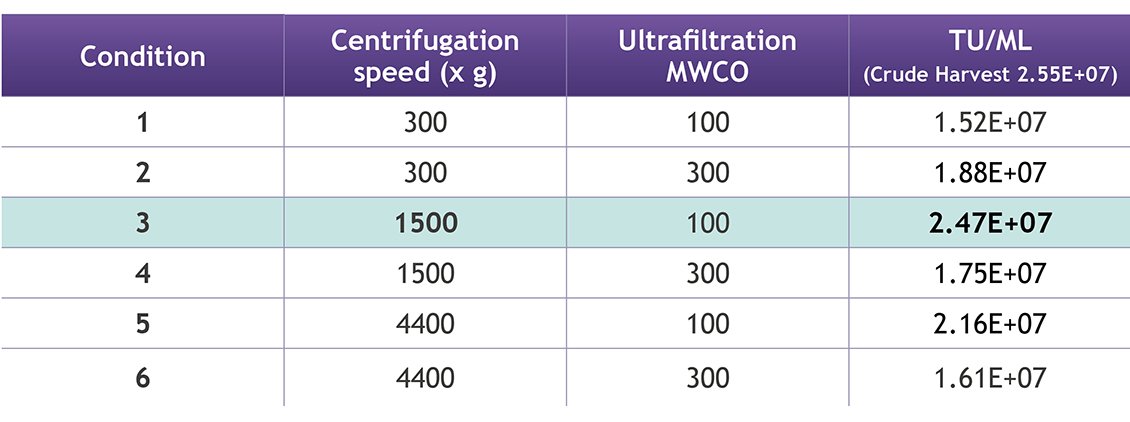
Table 1.
3. LentiHERO® – A Speedy Highly Efficient Screening Tool: The Nereus LentiHERO® spin column allows charged-based purification of LVV samples, thereby providing a screening tool that is indicative of large-scale processes. Samples of suspension feedstock clarified under each of the six conditions were purified using Nereus LentiHERO® spin columns in a benchtop centrifuge. This method allowed for the preparation of six conditions in less than 25 minutes (figure 2a). Conditions 3 and 5 resulted in the highest TU recovery (figure 2b).
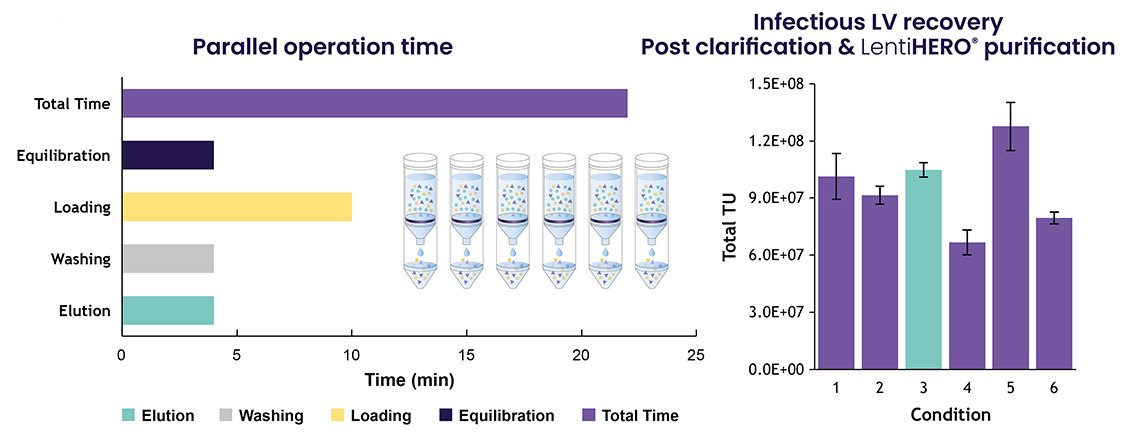
Figure 2a and 2b.
4. Minimal Effect on Optimized Clarification Step on Lentiviral Recovery: Suspension feedstock processed under Condition 3 and adherent feedstock were both purified with LentiHERO®. LVV recoveries were similar for both physical and infectious titers (figure 3a and b).
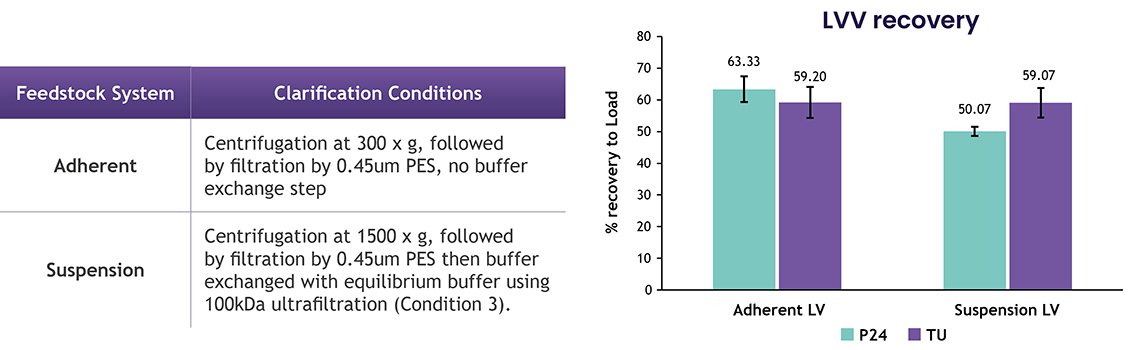
Figure 3a and 3b.
5. High HCP Reduction: Suspension feedstock post-clarification contained high levels of HCP. Nereus LentiHERO® purification significantly reduced HCP and process-related dsDNA contamination from both adherent and suspension feedstocks (figure 4a and b). HCP levels were barely detectable in the eluate (figure 4c).
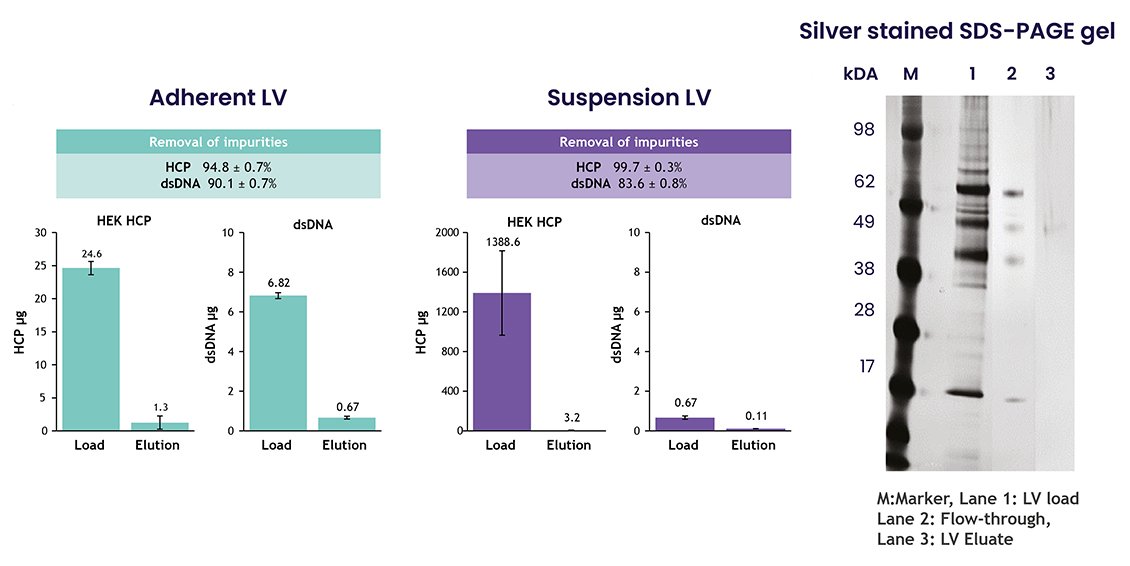
Figure 4a, 4b and 4c.
6. Lentiviral Structure Preservation: LV-specific p24 proteins, located in the capsid and often used to measure lentiviral titer, were identified in the eluted fractions (figure 5a). Transmission electron microscopy (TEM) images revealed the typical healthy morphologies of LVVs following LentiHERO® purification (figure 5b).
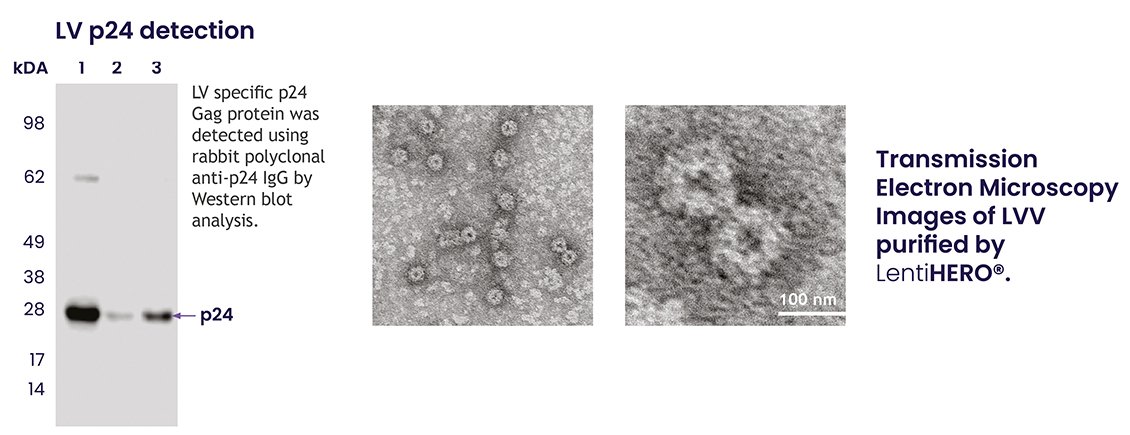
Figure 5a and 5b.
7. High Dynamic Binding Capacity of Nereus LentiHERO®: The study also demonstrated a high dynamic binding capacity (DBC) of Nereus LentiHERO® (figure 6) with recovery of physical particles (PP) and infectious particles (Table 2). This indicates the significant potential of the composite nanofiber adsorbent for processing at scale.
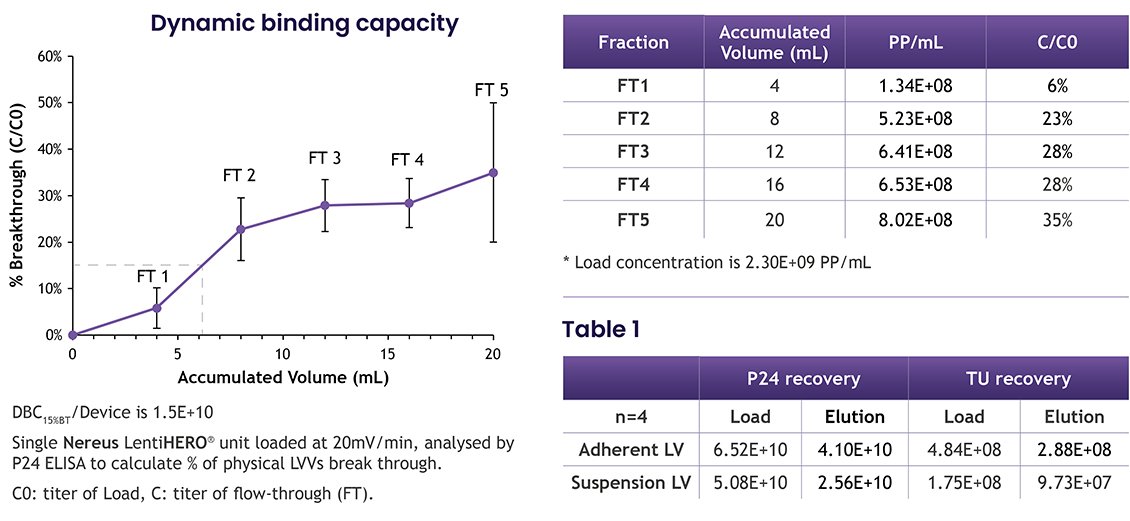
Figure 6 and table 2.
Key Findings
This study has revealed several key findings that have significant implications for the development of LVV-based therapeutic assets:
Efficient Sample Preparation Workflow: Nereus LentiHERO® purification, when combined with an optimized clarification process, offers an efficient sample preparation workflow for suspension feedstock.
Parallel Purification: Nereus LentiHERO® enables parallel purification of samples, facilitating scale-out screening studies.
High Dynamic Binding Capacity: The high dynamic binding capacity of Nereus LentiHERO® suggests its potential for large-scale processing, with a DBC of 1.25E+11 PP/mL, making it a powerful tool for downstream processing condition development.
Conclusion
The development of LVV-based therapeutic assets has faced significant challenges, particularly in sample preparation, which can impact pipeline progress. The introduction of Nereus LentiHERO®, as an efficient screening tool, and the optimization of the clarification process represent a significant step forward in overcoming these hurdles. This parallel purification strategy not only streamlines the processing of suspension feedstock but also maintains the integrity of LVVs and reduces contaminants, such as HCP. These findings hold promise for the advancement of gene therapy and bioprocessing on a larger scale, bringing us one step closer to more efficient and effective treatments for genetic diseases. The Nereus LentiHERO® device has been demonstrated as a robust tool for the “process compression” of lentiviral preparation. We have shown that the potential for process gains increases with the increased scale the purification device. As such, Astrea Bioseparations are committed building out the suite of LentiHERO® liquid chromatography compatible devices. Most recently, the 1 mL and 10 mL devices were launched at BPI Boston and are the natural evolution in scale from the Nereus device outlines in this study. These larger devices bring processing power to the user that is unparalleled.



